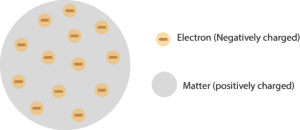
Thomson discovered that there was a particle smaller than an atom - the electron - through his work with cathode ray tubes. However, in 1897, the English physicist J. The work of Avogadro on the volumes of gases and Robert Brown on Brownian motion further supported this hypothesis.

This theory was established by Dalton and the atomists who stated that matter was made up of small indivisible particles known as elements which varied in their nature. The plum pudding model was the first model to suggest that atoms were not the smallest unit of matter, and that they may be made up of both positive and negatively charged subatomic particles.įor a long time, atoms were thought to be the smallest unit of matter. What did the plum pudding model suggest about atoms? Instead of a sea of positive charges surrounding negative charged particles, atoms are actually organized with positive and neutral particles at their core (the nucleus) surrounded by orbitals of electrons.ģ. While the plum pudding model was the first to suggest that atoms are made up of charged particles, the plum pudding model is not entirely correct. This work was later expanded upon by chemists throughout the 19th century and beyond to create the modern model of the atom, which arose after the rise and fall of many partial theories.ġ. The modern atomic model started to take shape with the work of Lavoisier and Dalton who formulated the concept of elements as unique iterations of atoms which can be combined in multiple ways to create a variety of substances. Refresher: The atomic model originated in the 5th century BC when Greek philosophers hypothesized that all matter was composed of indivisible particles.
